How Is Nitrogen Used In Plants And Animals
Abstract
Nitrogen, the most arable element in our atmosphere, is crucial to life. Nitrogen is constitute in soils and plants, in the water we drink, and in the air we breathe. It is too essential to life: a central building cake of DNA, which determines our genetics, is essential to plant growth, and therefore necessary for the food nosotros grow. Merely as with everything, remainder is key: too little nitrogen and plants cannot thrive, leading to low crop yields; but too much nitrogen can be toxic to plants, and tin as well damage our environment. Plants that practice not have enough nitrogen become yellowish and practise not grow well and tin have smaller flowers and fruits. Farmers can add together nitrogen fertilizer to produce better crops, but besides much can hurt plants and animals, and pollute our aquatic systems. Agreement the Nitrogen Bike—how nitrogen moves from the atmosphere to earth, through soils and back to the atmosphere in an endless Cycle—tin can help us grow healthy crops and protect our environment.
Introduction
Nitrogen, or N, using its scientific abbreviation, is a colorless, odorless element. Nitrogen is in the soil nether our feet, in the water we beverage, and in the air we breathe. In fact, nitrogen is the most abundant chemical element in World's atmosphere: approximately 78% of the atmosphere is nitrogen! Nitrogen is of import to all living things, including us. It plays a cardinal role in institute growth: too trivial nitrogen and plants cannot thrive, leading to depression crop yields; merely likewise much nitrogen can exist toxic to plants [1]. Nitrogen is necessary for our food supply, just backlog nitrogen can damage the surroundings.
Why Is Nitrogen Important?
The delicate rest of substances that is of import for maintaining life is an important area of inquiry, and the balance of nitrogen in the environment is no exception [ii]. When plants lack nitrogen, they get yellowed, with stunted growth, and produce smaller fruits and flowers. Farmers may add fertilizers containing nitrogen to their crops, to increment crop growth. Without nitrogen fertilizers, scientists estimate that nosotros would lose up to one third of the crops nosotros rely on for nutrient and other types of agriculture. But we need to know how much nitrogen is necessary for plant growth, because too much can pollute waterways, hurting aquatic life.
Nitrogen Is Cardinal to Life!
Nitrogen is a key chemical element in the nucleic acids Dna and RNA , which are the well-nigh important of all biological molecules and crucial for all living things. DNA carries the genetic information, which means the instructions for how to make up a life form. When plants exercise not get enough nitrogen, they are unable to produce amino acids (substances that contain nitrogen and hydrogen and make up many of living cells, muscles and tissue). Without amino acids, plants cannot make the special proteins that the plant cells need to grow. Without enough nitrogen, found growth is affected negatively. With too much nitrogen, plants produce excess biomass, or organic matter, such as stalks and leaves, only not enough root structure. In extreme cases, plants with very loftier levels of nitrogen captivated from soils can poison subcontract animals that eat them [3].
What Is Eutrophication and tin Information technology Be Prevented?
Excess nitrogen can also leach—or drain—from the soil into hush-hush water sources, or information technology can enter aquatic systems as above basis runoff. This excess nitrogen can build upwardly, leading to a process called eutrophication . Eutrophication happens when likewise much nitrogen enriches the water, causing excessive growth of plants and algae. Too much nitrogen tin can even cause a lake to turn vivid green or other colors, with a "bloom" of evil-smelling algae called phytoplankton (meet Effigy ane)! When the phytoplankton dies, microbes in the water decompose them. The process of decomposition reduces the amount of dissolved oxygen in the water, and can atomic number 82 to a "dead zone" that does not have enough oxygen to back up virtually life forms. Organisms in the dead zone die from lack of oxygen. These dead zones can happen in freshwater lakes and also in coastal environments where rivers full of nutrients from agricultural runoff (fertilizer overflow) flow into oceans [4].

- Figure ane - Eutrophication at a waste water outlet in the Potomac River, Washington, D.C.
- The water in this river, is bright green because information technology has undergone eutrophication, due to excess nitrogen and other nutrients polluting the h2o, which has led to increased phytoplankton and algal blooms, and then the water has get cloudy and tin turn unlike colors, such as green, yellowish, crimson, or dark-brown, depending on the algal blooms (Wikimedia Commons: https://commons.wikimedia.org/wiki/Category:Eutrophication#/media/File:Potomac_green_water.JPG).
Figure 2 shows the stages of Eutrophication (open up admission Wikimedia Commons image from https://commons.m.wikimedia.org/wiki/File:Eutrophicationmodel.svg).
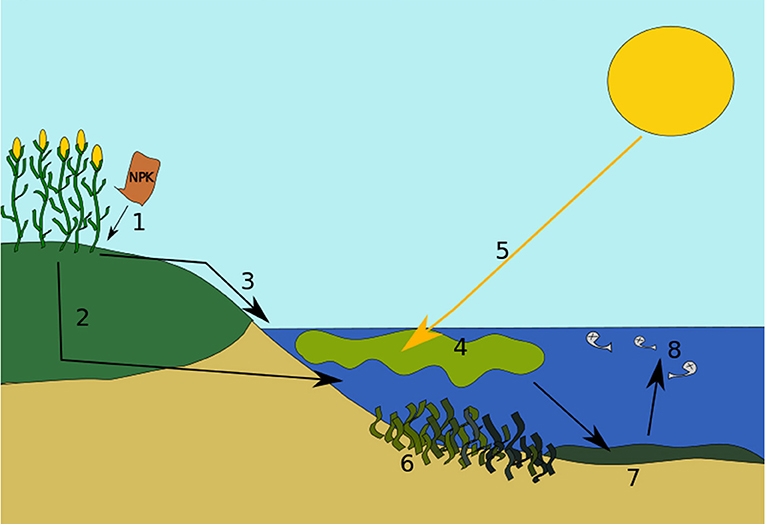
- Figure two - Stages of eutrophication.
- (1) Excess nutrients stop up in the soil and basis. (two) Some nutrients become dissolved in water and leach or leak into deeper soil layers. Somewhen, they go drained into a water body, such equally a lake or pond. (3) Some nutrients run off from over the soils and ground directly into the h2o. (four) The actress nutrients crusade algae to flower. (5) Sunlight becomes blocked by the algae. (six) Photosynthesis and growth of plants under the water will be weakened or potentially stopped. (7) Next, the algae bloom dies and falls to the bottom of the water body. And then, bacteria begin to decompose or interruption up the remains, and use upwards oxygen in the process. (8) The decomposition process causes the water to have reduced oxygen, leading to "expressionless zones." Bigger life forms like fish cannot exhale and dice. The water torso has at present undergone eutrophication.
Tin eutrophication be prevented? Yep! People who manage water resources can use different strategies to reduce the harmful effects of algal blooms and eutrophication of water surfaces. They can re-reroute excess nutrients away from lakes and vulnerable costal zones, use herbicides (chemicals used to kill unwanted plant growth) or algaecides (chemicals used to kill algae) to stop the algal blooms, and reduce the quantities or combinations of nutrients used in agricultural fertilizers, among other techniques [5]. But, it tin often be hard to notice the origin of the excess nitrogen and other nutrients.
Once a lake has undergone eutrophication, it is even harder to practise damage control. Algaecides can exist expensive, and they also do non correct the source of the trouble: the excess nitrogen or other nutrients that caused the algae bloom in the start place! Another potential solution is chosen bioremediation , which is the process of purposefully changing the nutrient spider web in an aquatic ecosystem to reduce or control the amount of phytoplankton. For example, water managers tin can introduce organisms that eat phytoplankton, and these organisms tin can aid reduce the amounts of phytoplankton, by eating them!
What Exactly Is the Nitrogen Cycle?
The nitrogen wheel is a repeating cycle of processes during which nitrogen moves through both living and non-living things: the temper, soil, water, plants, animals and leaner . In order to move through the different parts of the cycle, nitrogen must change forms. In the atmosphere, nitrogen exists as a gas (North2), but in the soils information technology exists as nitrogen oxide, NO, and nitrogen dioxide, NO2, and when used every bit a fertilizer, can be found in other forms, such every bit ammonia, NH3, which can be processed even further into a unlike fertilizer, ammonium nitrate, or NH4NO3.
In that location are 5 stages in the nitrogen cycle, and we volition now discuss each of them in turn: fixation or volatilization, mineralization, nitrification, immobilization, and denitrification. In this prototype, microbes in the soil plow nitrogen gas (N2) into what is chosen volatile ammonia (NH3), so the fixation procedure is chosen volatilization. Leaching is where certain forms of nitrogen (such every bit nitrate, or NO3) becomes dissolved in water and leaks out of the soil, potentially polluting waterways.
Phase 1: Nitrogen Fixation
In this phase, nitrogen moves from the atmosphere into the soil. Globe's atmosphere contains a huge puddle of nitrogen gas (N2). But this nitrogen is "unavailable" to plants, because the gaseous class cannot be used straight by plants without undergoing a transformation. To be used by plants, the Due north2 must be transformed through a process called nitrogen fixation. Fixation converts nitrogen in the atmosphere into forms that plants tin can absorb through their root systems.
A small amount of nitrogen can be fixed when lightning provides the energy needed for N2 to react with oxygen, producing nitrogen oxide, NO, and nitrogen dioxide, NOtwo. These forms of nitrogen then enter soils through pelting or snowfall. Nitrogen tin can likewise be fixed through the industrial procedure that creates fertilizer. This course of fixing occurs nether high rut and pressure level, during which atmospheric nitrogen and hydrogen are combined to form ammonia (NH3), which may and so exist processed further, to produce ammonium nitrate (NH4NOiii), a course of nitrogen that tin can be added to soils and used past plants.
Nigh nitrogen fixation occurs naturally, in the soil, by bacteria. In Figure iii (to a higher place), you can see nitrogen fixation and exchange of grade occurring in the soil. Some bacteria adhere to found roots and have a symbiotic (beneficial for both the plant and the bacteria) relationship with the plant [half-dozen]. The bacteria get free energy through photosynthesis and, in return, they fix nitrogen into a form the plant needs. The fixed nitrogen is then carried to other parts of the plant and is used to form plant tissues, and so the plant tin grow. Other leaner live freely in soils or h2o and tin can set nitrogen without this symbiotic relationship. These leaner tin can besides create forms of nitrogen that can be used past organisms.
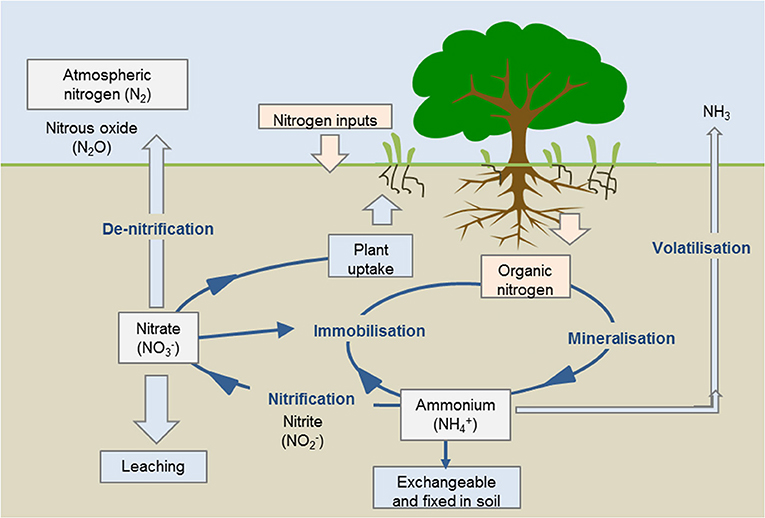
- Figure 3 - Stages of the nitrogen cycle.
- The Nitrogen Bike: Nitrogen cycling through the various forms in soil determines the amount of nitrogen available for plants to uptake. Source: https://world wide web.agric.wa.gov.au/soil-carbon/immobilisation-soil-nitrogen-heavy-stubble-loads.
Stage two: Mineralization
This stage takes identify in the soil. Nitrogen moves from organic materials, such equally manure or plant materials to an inorganic form of nitrogen that plants can use. Eventually, the establish's nutrients are used up and the found dies and decomposes. This becomes important in the second stage of the nitrogen wheel. Mineralization happens when microbes deed on organic textile, such as animal manure or decomposing plant or brute cloth and begin to catechumen information technology to a form of nitrogen that can be used by plants. All plants under cultivation, except legumes (plants with seed pods that split up in half, such as lentils, beans, peas or peanuts) get the nitrogen they require through the soil. Legumes get nitrogen through fixation that occurs in their root nodules, equally described above.
The outset form of nitrogen produced past the process of mineralization is ammonia, NH3. The NH3 in the soil so reacts with water to course ammonium, NH4. This ammonium is held in the soils and is available for use by plants that practice not get nitrogen through the symbiotic nitrogen fixing relationship described to a higher place.
Stage 3: Nitrification
The third phase, nitrification, besides occurs in soils. During nitrification the ammonia in the soils, produced during mineralization, is converted into compounds called nitrites, NO2 −, and nitrates, NO3 −. Nitrates tin be used by plants and animals that consume the plants. Some bacteria in the soil can plough ammonia into nitrites. Although nitrite is not usable past plants and animals directly, other leaner can alter nitrites into nitrates—a form that is usable by plants and animals. This reaction provides energy for the bacteria engaged in this process. The leaner that we are talking about are called nitrosomonas and nitrobacter. Nitrobacter turns nitrites into nitrates; nitrosomonas transform ammonia to nitrites. Both kinds of bacteria can act just in the presence of oxygen, O2 [7]. The procedure of nitrification is important to plants, as it produces an extra stash of available nitrogen that can be captivated by the plants through their root systems.
Stage iv: Immobilization
The quaternary stage of the nitrogen wheel is immobilization, sometimes described as the contrary of mineralization. These two processes together control the amount of nitrogen in soils. Just like plants, microorganisms living in the soil require nitrogen every bit an energy source. These soil microorganisms pull nitrogen from the soil when the residues of decomposing plants do not contain enough nitrogen. When microorganisms take in ammonium (NH4 +) and nitrate (NOthree −), these forms of nitrogen are no longer available to the plants and may cause nitrogen deficiency, or a lack of nitrogen. Immobilization, therefore, ties up nitrogen in microorganisms. However, immobilization is of import because it helps control and rest the corporeality of nitrogen in the soils by tying it up, or immobilizing the nitrogen, in microorganisms.
Stage 5: Denitrification
In the 5th phase of the nitrogen cycle, nitrogen returns to the air every bit nitrates are converted to atmospheric nitrogen (N2) past bacteria through the process we phone call denitrification. This results in an overall loss of nitrogen from soils, as the gaseous form of nitrogen moves into the atmosphere, back where nosotros began our story.
Nitrogen Is Crucial for Life
The cycling of nitrogen through the ecosystem is crucial for maintaining productive and healthy ecosystems with neither likewise much nor likewise little nitrogen. Plant product and biomass (living material) are express by the availability of nitrogen. Understanding how the plant-soil nitrogen cycle works tin can help us brand better decisions well-nigh what crops to grow and where to abound them, so we have an acceptable supply of nutrient. Knowledge of the nitrogen cycle can also help us reduce pollution caused past adding too much fertilizer to soils. Certain plants can uptake more nitrogen or other nutrients, such as phosphorous, some other fertilizer, and tin even exist used every bit a "buffer," or filter, to foreclose excessive fertilizer from inbound waterways. For instance, a study done by Haycock and Pinay [8] showed that poplar trees (Populus italica) used as a buffer held on to 99% of the nitrate entering the underground h2o flow during winter, while a riverbank zone covered with a specific grass (Lolium perenne Fifty.) held upward to 84% of the nitrate, preventing it from entering the river.
As you have seen, not plenty nitrogen in the soils leaves plants hungry, while too much of a expert thing can exist bad: backlog nitrogen can poison plants and even livestock! Pollution of our water sources by surplus nitrogen and other nutrients is a huge trouble, as marine life is existence suffocated from decomposition of dead algae blooms. Farmers and communities need to work to improve the uptake of added nutrients by crops and treat creature manure waste matter properly. We likewise need to protect the natural plant buffer zones that tin take up nitrogen runoff before it reaches h2o bodies. Simply, our current patterns of clearing copse to build roads and other structure worsen this trouble, because there are fewer plants left to uptake excess nutrients. We need to exercise further research to decide which establish species are best to grow in coastal areas to take upward excess nitrogen. We likewise need to notice other ways to fix or avoid the trouble of excess nitrogen spilling over into aquatic ecosystems. By working toward a more complete agreement of the nitrogen bicycle and other cycles at play in Earth's interconnected natural systems, we tin improve sympathize how to better protect Earth's precious natural resources.
Glossary
DNA: ↑ Deoxyribonucleic acid, a self-replicating cloth which is present in nigh all living organisms as the main component of chromosomes, and carrier of genetic information.
RNA: ↑ Ribonucleic acid, a nucleic acid nowadays in all living cells, acts as a messenger conveying instructions from Deoxyribonucleic acid.
Eutrophication: ↑ Excessive amount of nutrients (such equally nitrogen) in a lake or other body of water, which causes a dumbo growth of aquatic plant life, such as algae.
Phytoplankton: ↑ Tiny, microscopic marine algae (also known every bit microalgae) that require sunlight in order to grow.
Bioremediation: ↑ Using other microorganisms or tiny living creatures to eat and break downwardly pollution in order to make clean a polluted site.
Bacteria: ↑ Microscopic living organisms that usually comprise only one cell and are institute everywhere. Leaner can cause decomposition or breaking down, of organic material in soils.
Leaching: ↑ When a mineral or chemical (such equally nitrate, or NOthree) drains away from soil or other basis fabric and leaks into surrounding area.
Legumes: ↑ A member of the pea family: beans, lentils, soybeans, peanuts and peas, are plants with seed pods that dissever in half.
Microorganism: ↑ An organism, or living affair, that is too tiny to be seen without a microscope, such as a bacterium.
Conflict of Involvement Statement
The author declares that the inquiry was conducted in the absence of whatsoever commercial or financial relationships that could be construed as a potential conflict of involvement.
References
[1] ↑ Britto, D. T., and Kronzuker, H. J. 2002. NH4 + toxicity in higher plants: a critical review. J. Plant Physiol. 159:567–84. doi: ten.1078/0176-1617-0774
[2] ↑ Weathers, Yard. C., Groffman, P. K., Dolah, E. V., Bernhardt, E., Grimm, North. B., McMahon, K., et al. 2016. Frontiers in ecosystem ecology from a community perspective: the future is boundless and vivid. Ecosystems 19:753–70. doi: 10.1007/s10021-016-9967-0
[iii] ↑ Brady, Northward., and Weil, R. 2010. "Nutrient cycles and soil fertility," in Elements of the Nature and Properties of Soils, 3rd Edn, ed 5. R. Anthony (Upper Saddle River, NJ: Pearson Education Inc.), 396–420.
[4] ↑ Foth, H. 1990. Chapter 12: "Plant-Soil Macronutrient Relations," in Fundamentals of Soil Science, eighth Edn, ed John Wiley and Sons (New York, NY: John Wiley Company), 186–209.
[5] ↑ Chislock, G. F., Doster, E., Zitomer, R. A., and Wilson, A. E. 2013. Eutrophication: causes, consequences, and controls in aquatic ecosystems. Nat. Educ. Knowl. 4:10. Bachelor online at: https://world wide web.nature.com/scitable/knowledge/library/eutrophication-causes-consequences-and-controls-in-aquatic-102364466
[six] ↑ Peoples, M. B., Herridge, D. F., and Ladha, J. Thousand. 1995. Biological nitrogen fixation: an efficient source of nitrogen for sustainable agricultural production? Establish Soil 174:three–28. doi: ten.1007/BF00032239
[vii] ↑ Manahan, S. E. 2010. Environmental Chemistry, 9th Edn. Boca Raton, FL: CRC Press, 166–72.
[eight] ↑ Haycock, N. E., and Pinay, G. 1993. Groundwater nitrate dynamics in grass and poplar vegetated riparian buffer strips during the winter. J. Environ. Qual. 22:273–eight. doi: x.2134/jeq1993.00472425002200020007x
Source: https://kids.frontiersin.org/articles/10.3389/frym.2019.00041
Posted by: flemingyourejough.blogspot.com

0 Response to "How Is Nitrogen Used In Plants And Animals"
Post a Comment